
Origin® Two

For healthcare practitioners and device manufacturers, the Stratasys® Origin® Two due to its and open material platform enables the production of parts suitable for body contact.
-
P3™ DLP (Programmable Photopolymerization Digital Light Processing) Printing Technology
-
192 x 108 x 370 mm Build Volume
-
+/-50 μm (25 μm – 200 μm layers with OpenAM™) Layer Thickness
-
13+ different materials Material Compatibility
-
5K projector with 38.5 μm pixel size Light Engine
High Level of Detail
For dental and medical applications, the Origin® Two's remarkable precision and surface polish are exceptional. It comes with a 5K projector with 38.5 μm pixel size and tight 2.5 µm standard deviation projector uniformity. It can create products with an injection-molding-quality finish right out of the printer with precision up to ±50 µm and surface roughness as low as 2μm Ra.
Creating complex medical equipment, dental implants, and anatomical models that need great dimensional precision requires this degree of detail. For example, the accuracy of the Origin® Two guarantees that each device can be customized to the individual's distinct anatomy in the manufacturing of dental aligners or hearing aids, where patient comfort and fit are crucial.
In addition to improving patient outcomes, this capability streamlines the production workflow in dental and medical offices by lowering the need for laborious post-processing.
Open Material Platform
The Origin® Two's open material platform supports a wide range of high-performance photopolymers, including biocompatible resins suitable for continuous skin contact and short-term mucous membrane contact. This adaptability lets medical personnel build biocompatible devices, surgical guides, and custom trays utilizing a single printer.
The manufacturing of autoclavable medical instruments is also made possible by the utilization of materials with high heat deflection temperatures up to 300°C. This feature is particularly valuable in hospital settings where sterilization is critical.
By enabling the use of specially formulated resins suited to particular applications, such as resins with antimicrobial qualities or those made to replicate particular tissue properties for more precise prototyping and testing, the open material platform helps medical device companies innovate.
Outstanding Capabilities
Medical, dental, veterinary or pharmaceutical applications the Origin® Two can do it all. Because the printer can create extremely exact parts for various applications, they can be fitted more precisely, potentially saving patients more chair time and enhancing treatment results.
When combined with materials such as the Biocompatible Origin DM200 for example, the Origin® Two simplifies the manufacturing process for a variety of dental goods. It reduces operating time and material waste by enabling the production of dental models, surgical guides, denture try-ins, and bespoke trays utilizing a single resin tray. In busy dental labs and clinics where quick turnaround times are crucial, this efficiency is especially beneficial.
Furthermore, the creation of personalized surgical guidelines for dental implant operations improves the accuracy of these intricate procedures, which may benefit patients in the long run.
Designed for High Regulatory Standards
The Origin® Two is suitable for industrial and medical applications because it was created with safety and regulatory compliance in mind. Its door is fitted with a NRTL interlock, which improves facility and operator safety compliance. The printer has CE, UKCA, FCC, and RCM certifications; NRTL certification is still pending. This conformance guarantees that the Origin Two satisfies stringent safety and electromagnetic compatibility standards in many geographical areas.
The printer also encourages the use of materials that meet ISO 10993-5/-10 standards for biocompatibility and UL certifications for flame resistance.
These certifications, combined with the printer's ability to produce parts with medical-grade quality, make the Origin® Two an ideal choice for manufacturing biocompatible medical devices and other applications requiring high regulatory standards.
Sophisticated Software
The Origin® Two is a perfect match for medical device companies involved in low-volume, high-mix production scenarios or rapid prototyping of new devices because of its sophisticated software and real-time process monitoring, which guarantee consistent part quality and traceability, which are essential for fulfilling regulatory requirements in the medical industry.
Every part satisfies the exacting quality standards needed for medical applications because of the real-time monitoring capabilities, which enable the prompt detection and repair of any print irregularities. This degree of control and traceability is crucial for academic medical facilities and research organizations to preserve the integrity of clinical trials and research investigations.
Having a reliable system promotes the ability to produce small batches of customized medical devices or research tools quickly and cost-effectively to accelerate the pace of medical innovation and research.
Applications
Anatomical Models for Surgical Planning
Produce 3D-printed anatomical models of patient organs to simulate surgeries, allowing surgeons to practice procedures like resections and stitching with precision, ultimately reducing operation time and improving outcomes.
Custom Prosthetics
Highly functional prosthetic limbs can be prototyped with biocompatible materials, such as a prosthetic arm equipped with electromyography sensors to enable movement based on muscle signals, providing both strength and functionality.
Medical Device Prototypes
Create prototypes of surgical instruments and implantable devices with injection-molding-like surface quality, ensuring tight tolerances and superior performance during testing and validation phases.
Dental Applications
Produce biocompatible dental surgical guides with high precision, enabling dentists to perform accurate implant placements while reducing patient discomfort and procedural errors. Also create dental models, denture try-ins, and custom trays among many other possibilities.
Compatible Materials


Evonik INFINAM® TI 5400 L

Evonik INFINAM® RG 2000 L

Evonik INFINAM® RG 7100 L

Evonik INFINAM® RG 3101 L

Evonik INFINAM® ST 6100 L
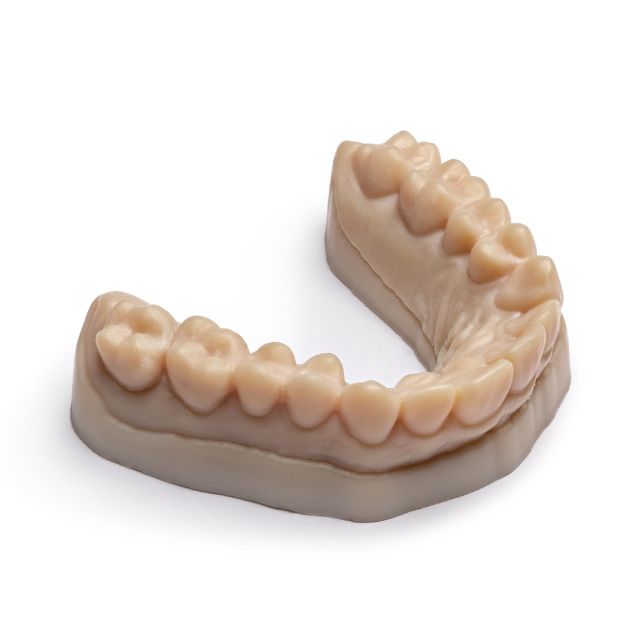
Stratasys® Biocompatible Origin DM200™

Stratasys® LOCTITE® MED413™

Nexa3D® xMED412

LOCTITE 3D IND402 A70 High Rebound

LOCTITE 3D IND405 HDT50 High Elongation

Stratasys® LOCTITE® Dura56

Stratasys® LOCTITE® 3D IND403 High Modulus

Stratasys® LOCTITE® 3D 3955 FST

LOCTITE 3D 3843 HDT60 High Toughness

Stratasys® LOCTITE® 3D 3172 High Impact

Ultracur3D® ST 45

Ultracur3D® RG 3280

Nexa3D® keyprint® KeyGuide®
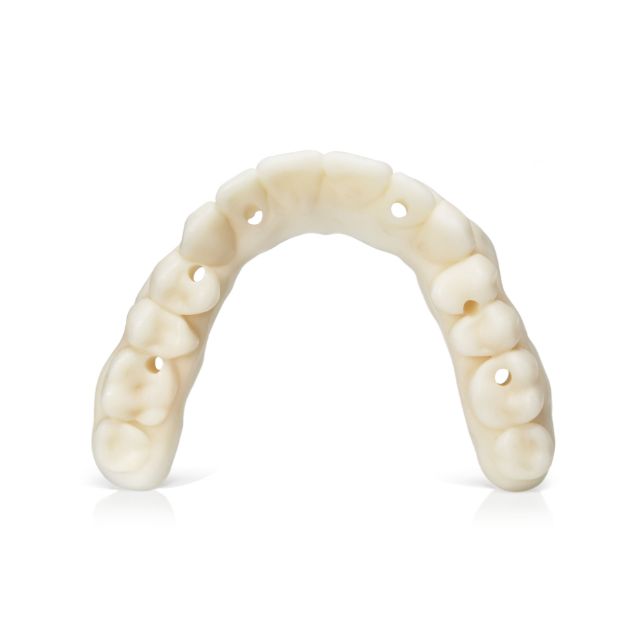
Stratasys® Keystone Industries KeyDenture Try-In™
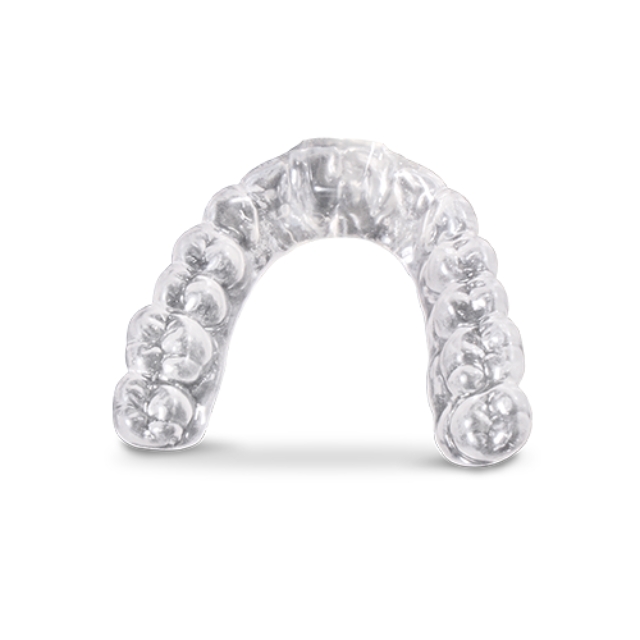
Nexa3D® keyprint® KeySplint Hard®
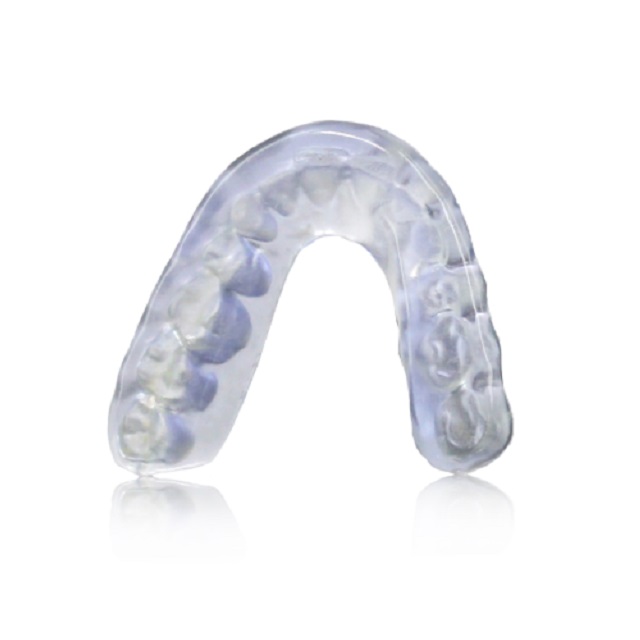
Nexa3D® keyprint® KeySplint Soft™
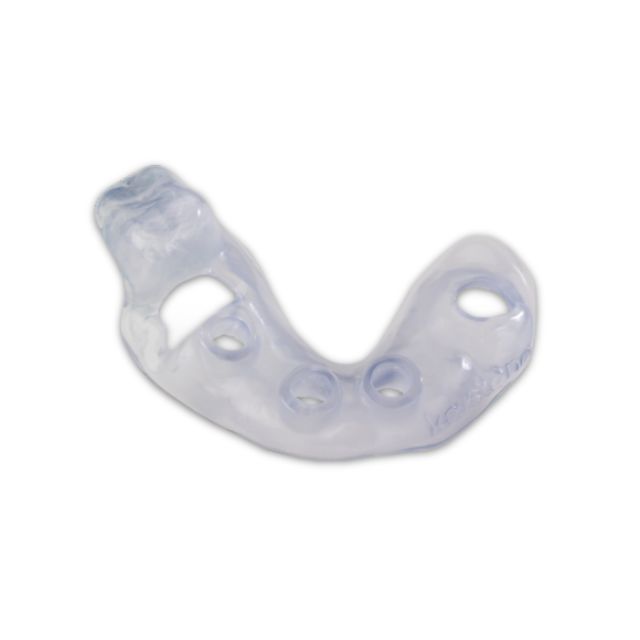
Stratasys® Keystone Industries KeyGuide®

Nexa3D® keyprint® KeyTray™

Stratasys® Keystone Industries KeySplint Hard®
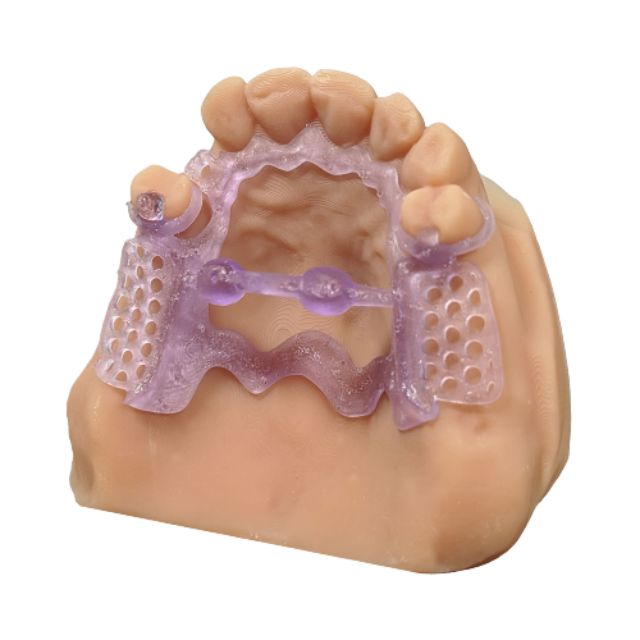
Stratasys® Keystone Industries KeyCast®
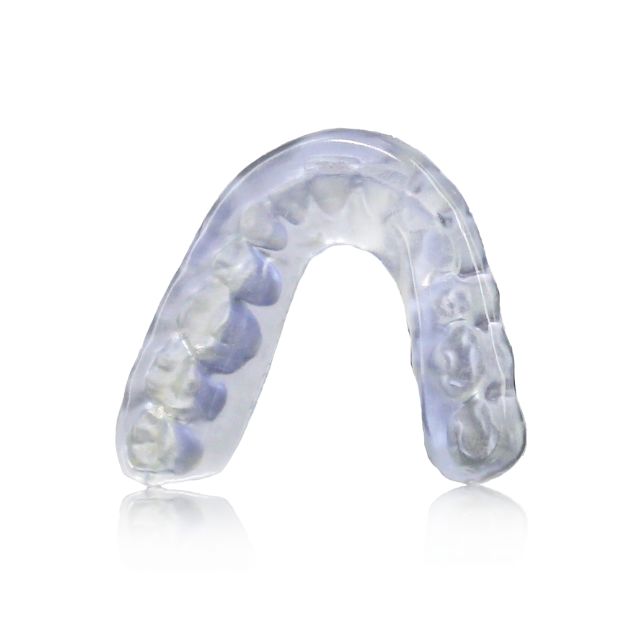
Stratasys® Keystone Industries KeySplint Soft®
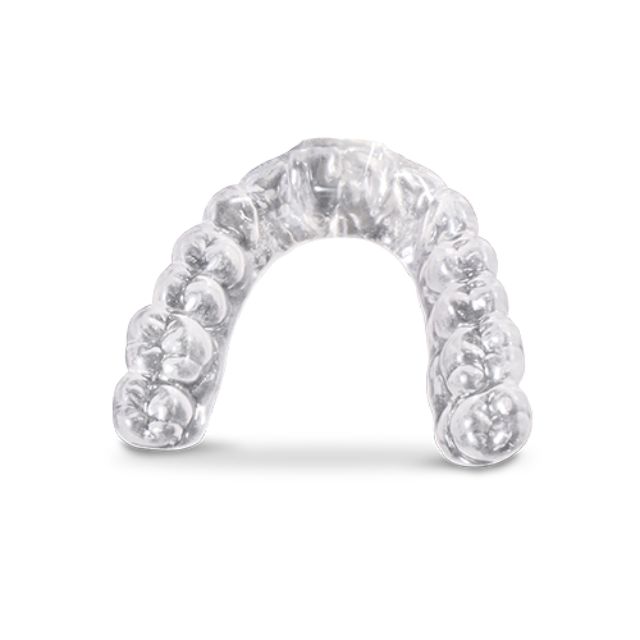
Stratasys® Keystone Industries KeySplint Soft® Clear

Stratasys® Keystone Industries KeyMask®
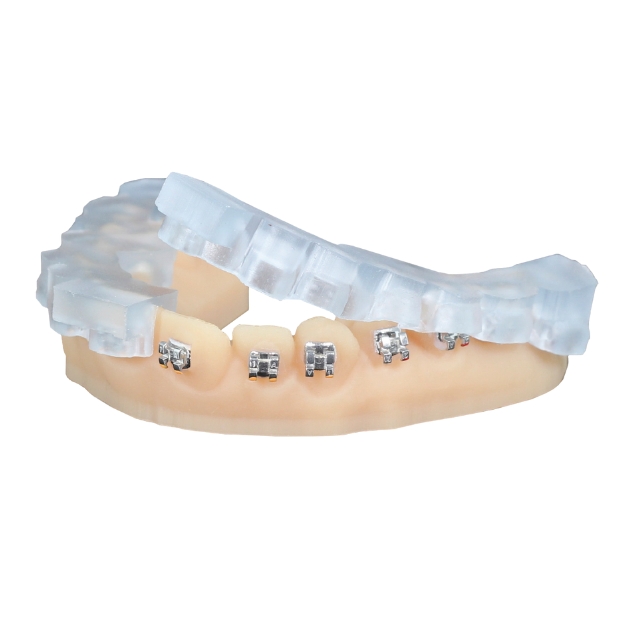
Nexa3D® keyprint® KeyOrtho IBT™
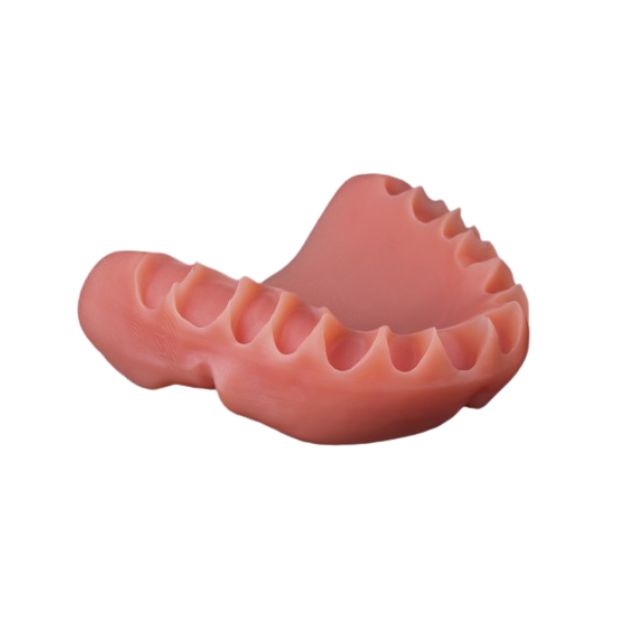
Stratasys® Dreve FotoDent® denture 385/405 nm

Stratasys® P3™ Deflect™ 190 ESD

Stratasys® Evonik INFINAM® P3™ Deflect™ 120

Stratasys® P3™ Stretch IND475™

Stratasys® P3™ Stretch™ 80





